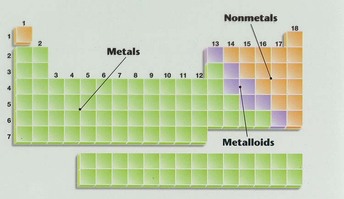
Here you can learn about Metals, Metalloids, and Non-Metals!
The following will list characteristics, definitions, important vocabulary, the elements in each group, and more!
The following will list characteristics, definitions, important vocabulary, the elements in each group, and more!

METALS-
-Definition: Elements used to conduct heat and electricity.
Characteristics-
-Metals are commonly solids at room temperature. --Lots of Metals shine.
-Metals conduct heat and electricity
-Metals are very Ductile
What are the elements in this group?
- There are different types of metals, Alkali Metals, Alkaline Earth Metals, and Transition Metals.
Alkali Metals-
-Characteristics- They are group 1 in the metal family and have 1 valance electron.
- They are soft, highly reactive, and silver in color.
- Some elements in this group are used to make, Metal Hydroxide and Hydrogen gas.
- They act very viciously towards air and water.
-The elements in Alkali Metals- Lithium, Sodium, Potassium, Rubidium, Caesium, and, Francium.
Alkaline Earth Metals-
-Characteristics- They are group 2 in the metal family.
- They have 2 valance electrons.
- They have a low density
- High melting point
- When made into fire, these elements change different colors.
- The elements in Alkaline Earth Metals- Berylium, Magnesium, Calcium, Strontium, Barium, and Radium.
Transition Metals
-Characteristics- They are groups 3-12 in the Metal family.
- They each give off different amount of valance electrons.
- They have high melting AND Boiling points.
- They have a high density.
-Elements in Transition Metals- Iron, Nickle, Copper, Silver, Gold, and ect. There are a total of 30 elements.
-Definition: Elements used to conduct heat and electricity.
Characteristics-
-Metals are commonly solids at room temperature. --Lots of Metals shine.
-Metals conduct heat and electricity
-Metals are very Ductile
What are the elements in this group?
- There are different types of metals, Alkali Metals, Alkaline Earth Metals, and Transition Metals.
Alkali Metals-
-Characteristics- They are group 1 in the metal family and have 1 valance electron.
- They are soft, highly reactive, and silver in color.
- Some elements in this group are used to make, Metal Hydroxide and Hydrogen gas.
- They act very viciously towards air and water.
-The elements in Alkali Metals- Lithium, Sodium, Potassium, Rubidium, Caesium, and, Francium.
Alkaline Earth Metals-
-Characteristics- They are group 2 in the metal family.
- They have 2 valance electrons.
- They have a low density
- High melting point
- When made into fire, these elements change different colors.
- The elements in Alkaline Earth Metals- Berylium, Magnesium, Calcium, Strontium, Barium, and Radium.
Transition Metals
-Characteristics- They are groups 3-12 in the Metal family.
- They each give off different amount of valance electrons.
- They have high melting AND Boiling points.
- They have a high density.
-Elements in Transition Metals- Iron, Nickle, Copper, Silver, Gold, and ect. There are a total of 30 elements.
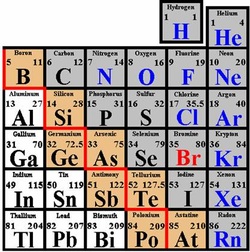
METALLOIDS-
Definition: An element whose properties are intermediate between those of metals and solid non-metals.
Characteristics- Elements found along the stair step line, that distinguishes metals from non-metals.
What are the elements in this group?
There are different types of Metalloids, Carbon family, Semiconductor, and Oxygen family.
The Carbon Family-
Characteristics- The Carbon Family is group 14. They have 4 valance electrons.
- It is called the "tin city".
- They are not very reactive.
- They occur in nature.
Elements in the Carbon Family- Carbon, Silicon, Germiniaum, Tin, Lead, and Fleromiu.
Semiconductor-
Characteristics- They have 3 valance electrons.
- They are groups 13-15.
- It is a class of crystalline solids.
- They have a high degree of chemical purity.
Elements in the Semiconductors- Carbon, Silicon, Germanium, and Antimony.
Oxygen Family-
Characteristics- They have 6 valance electrons.
- It is group 16.
- Oxygen and Sulfur are the most common two elements in this group.
- It is also called the "Chalcogens".
Elements in this family- Oxygen, Sulfur, Selenium, Tellurium, and Polonium.
Definition: An element whose properties are intermediate between those of metals and solid non-metals.
Characteristics- Elements found along the stair step line, that distinguishes metals from non-metals.
What are the elements in this group?
There are different types of Metalloids, Carbon family, Semiconductor, and Oxygen family.
The Carbon Family-
Characteristics- The Carbon Family is group 14. They have 4 valance electrons.
- It is called the "tin city".
- They are not very reactive.
- They occur in nature.
Elements in the Carbon Family- Carbon, Silicon, Germiniaum, Tin, Lead, and Fleromiu.
Semiconductor-
Characteristics- They have 3 valance electrons.
- They are groups 13-15.
- It is a class of crystalline solids.
- They have a high degree of chemical purity.
Elements in the Semiconductors- Carbon, Silicon, Germanium, and Antimony.
Oxygen Family-
Characteristics- They have 6 valance electrons.
- It is group 16.
- Oxygen and Sulfur are the most common two elements in this group.
- It is also called the "Chalcogens".
Elements in this family- Oxygen, Sulfur, Selenium, Tellurium, and Polonium.

NON-METALS
Definition: An element not having the character of a metal, as Carbon or Nitrogen.
Characteristics-
- High ionization energies.
- Generally poor conductors of heat and electricity.
- Solid non-metals, are usually brittle.
- Most non- Metals have the ability to gain electrons.
What are the elements in this group?
- There are different type of Non-Metals, Halogens, Noble Gases, and the Boron Family.
The Boron Family-
Characteristics- This is group 13.
- They have 3 valance electrons.
- Only one member of this family is a Metalloid. Which is Boron.
The elements in this group- Boron, Aluminum, Gallium, Indium, and Thallium.
Noble Gases-
Characteristics- They are group 18.
- They are unreactive.
- They are gases are room temperature.
- They have 8 valance electrons.
Elements in this group: Neon, Argon, Kripton, Xenon, and Helium.
Halogens-
Characteristics- They are in row 7 on the Periodic table.
- They have 7 valance electrons.
- They are extremely reactive.
- They can be found in many minerals in seawater.
Elements in this group: Fluorine, Chlorine, Bromine, Iodine, and Astatine.
Definition: An element not having the character of a metal, as Carbon or Nitrogen.
Characteristics-
- High ionization energies.
- Generally poor conductors of heat and electricity.
- Solid non-metals, are usually brittle.
- Most non- Metals have the ability to gain electrons.
What are the elements in this group?
- There are different type of Non-Metals, Halogens, Noble Gases, and the Boron Family.
The Boron Family-
Characteristics- This is group 13.
- They have 3 valance electrons.
- Only one member of this family is a Metalloid. Which is Boron.
The elements in this group- Boron, Aluminum, Gallium, Indium, and Thallium.
Noble Gases-
Characteristics- They are group 18.
- They are unreactive.
- They are gases are room temperature.
- They have 8 valance electrons.
Elements in this group: Neon, Argon, Kripton, Xenon, and Helium.
Halogens-
Characteristics- They are in row 7 on the Periodic table.
- They have 7 valance electrons.
- They are extremely reactive.
- They can be found in many minerals in seawater.
Elements in this group: Fluorine, Chlorine, Bromine, Iodine, and Astatine.
